Prevent The Spread Of Disease In Irrigation Water
 Phytophthora and Pythium are water molds that are difficult to control, especially under warm, wet conditions. Thick-walled oospores survive between crops on plant containers, benches, floors, and in potting media or soil.
Phytophthora and Pythium are water molds that are difficult to control, especially under warm, wet conditions. Thick-walled oospores survive between crops on plant containers, benches, floors, and in potting media or soil.
Spores include lemon-shaped sporangia, thick-walled chlamydospores, and swimming zoospores. The lemon-shaped spores called sporangia form on the surface of roots, crowns, and other plant tissues. Sporangia may be dislodged from infected plants by irrigation water, drainage water, or windblown rain. In water, sporangia release many swimming spores called zoospores, which spread the disease.
Pythium And Phytophthora: Water Mold Cousins
Pythium nibbles the feeding roots of plants, resulting in stunted growth and death. It can be introduced to the greenhouse via plant plugs or other prefinished plant material, or it can hibernate on dirty plant containers, benches, and walkways.
Although Pythium can be a problem on many annuals and perennials, it seems to favor geraniums and poinsettias. Sanitary conditions that favor good plant growth and minimize stress make the plant less vulnerable to attack by a root rot.
Two types of Phytophthora (Phytophthora nicotianae and P. drechsleri) are usually found in floriculture crops and can cause root, crown, and foliar blights. Symptoms include brown-black cankers at the soil line and diseased roots. P. nicotianae can infect snapdragon, fuchsia, verbena, bacopa, vinca, African violet, and dusty miller, while. P. drechsleri may infect poinsettias.
Recirculating Irrigation Water Can Spread Pythium and Phytophthora
Phytophthora spread occurs from zoospores that move through water. Disease occurrence within nurseries and greenhouses often follows drainage patterns, but windblown rain may allow Phytophthora to spread across a nursery. The cycle of plant infection and zoospore production occurs many times within a single growing season. Thus, low levels of infection early in the season may result in a rapid epidemic, if the disease is not immediately controlled.
Many physical and chemical treatment options have been proposed to disinfect contaminated irrigation, including treatment with substances (chlorine, ozone, surfactants, antimicrobial compounds, biological control agents, ultraviolet light, and sedimentation), and physical removal (filtration).
Treatment Methods For Contaminated Irrigation Water
Recycling irrigation water and recirculating nutrient solutions are beneficial, but can also increase the spread of waterborne pathogens. There are several methods currently available for disinfesting contaminated irrigation water, including the use of antimicrobial compounds such as copper and hydrogen peroxide.
Commercial algaecides contain copper sulfate, chelated copper, or sodium carbonate peroxyhydrate as their active ingredients. Previous studies show that increasing copper ion concentrations in nutrient solutions reduces disease caused by some Phytophthora spp.
Without adequate treatment options for irrigation water, water mold pathogens can continue to cause significant crop losses and reduce floriculture crop quality. One effective strategy to prevent crop losses is to limit pathogen numbers in critical transmission routes or points within a production system, such as in irrigation water. Sand filtration has been proposed as an inexpensive treatment to remove the water molds, including Pythium and Phytophthora, from contaminated water, but variable results have been observed. Spores of water molds are negatively charged, and interactions between spores and the sand filter surface are influenced by the physical and chemical properties of the water and sand.
Some greenhouse irrigation systems already have a filtration unit that uses a screen or paper filters to remove large-sized soil particles and plant debris to keep the micro-emitters of irrigation lines clear. These installed filtration units are not likely to be effective to remove water mold spores, which are much smaller in size.
Filtration Research At Michigan State University
A preliminary greenhouse filtration experiment tested sand filtration media containing positively charged, nano-sized iron-oxide particles embedded on the surface. Results showed that Phytophthora zoospores were readily attached to the synthesized, nano-sized, iron-oxide surface coating of filter sand. The removal of motile zoospores was significantly less than that of the encysted zoospores. The results suggest that through controlling parameters of zoospores (e.g., encystment), solution chemistry (e.g., ion type), or filter media type, zoospore removal can be optimized.
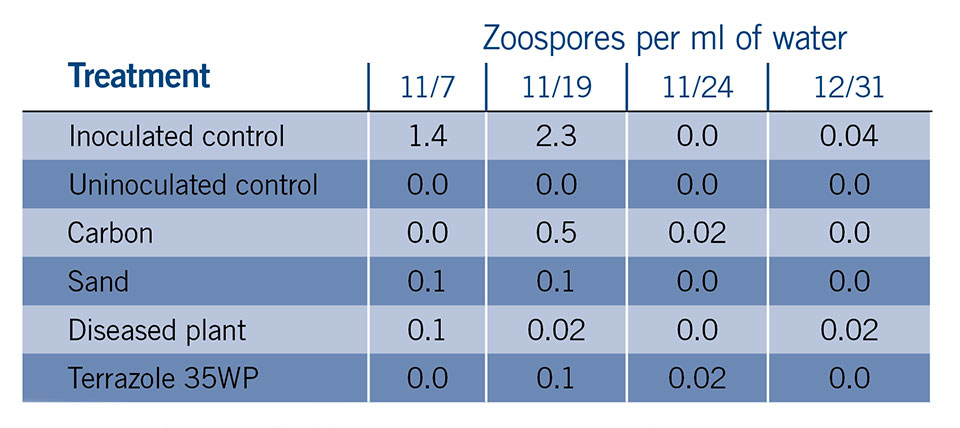
Novel filter media are being tested in small-scale filter units in a greenhouse environment with spores of Pythium aphanidermatum. A trial tested poinsettias that were (A) untreated uninoculated or (B) untreated inoculated, and compared them with inoculated plants irrigated with systems containing (C) a sand filter or (D) an activated carbon filter, and inoculated plants irrigated with (E) an unfiltered system with the labeled rate of Terrazole 35WP added to the water or (F) an unfiltered system that had diseased plants.
Zoospores were added to the pre-filter tanks and were passed through the filters and irrigated onto the ebb-and-flow bench tops to initiate the experiment. Plants were irrigated twice daily for 15 minutes to increase disease pressure. Water was sampled from the holding tanks during the course of the study to determine the concentration of zoospores in solution (Table 1, see slideshow).
[blackoutgallery id=”128650″]
The trial concluded after two and a half months. The inoculated control displayed severe root rot and stunting (Figure 1, see slideshow). The sand filter, Terrazole treatment, carbon filter, and diseased plant treatment all had significantly higher foliar fresh weight and plant height than the inoculated control treatment. These results suggest filtration may be useful in a greenhouse setting.