Unclog Drip Emitters In Your Greenhouse
If irrigation lines, drippers, or mist emitters are clogging in your greenhouse, one of three main water contaminants (minerals, particles, or biofilm), shown in Table 1 (see slideshow), are the most likely cause.
Let’s look at a real-world example from a leading citrus nursery grower, Himrod Citrus Nursery, in Florida. The grower built a 25,000-square-foot greenhouse to produce high-quality transplants, free of citrus greening and canker diseases (Figure 1, see slideshow). The company was using well water, water-soluble fertilizer, and a line cleaner. It had placed drip irrigation into 2.7-liter pots to avoid wetting foliage and transmitting plant pathogens. The grower also installed a 100-micron, central stainless-steel screen filter (Figure 2E, see slideshow) and smaller 75-micron, plastic spin-clean screen filters in the greenhouse.
Issues rapidly arose with the drippers clogging, and individual plants on benches started drying out (Figure 2, see slideshow). At first, the clogs in the drippers mainly affected irrigation uniformity. However, the problem progressed to the point where many of the drippers were entirely plugged.
Himrod’s employees constantly monitored for dry trees and replaced clogged drippers. Workers had to hand water trees (at a labor cost of around $1,000/week), and even a temporary sprinkler system was clogging. The filters needed manual cleaning every two weeks with high-pressure water and a nylon brush to remove the brown residue. After thoroughly removing sediment from the lines with a cleaning agent, the drippers would clog up again within weeks. Owner Aaron Himrod quickly concluded that the existing state of affairs was completely unsustainable.
Water Testing To Diagnose The Problem
The first step to help diagnose this problem was to sample and analyze water at the well-head and out in the greenhouse. The well water had a pH of 6.1 (ideal for crop growth). The electrical conductivity (EC) of 0.1 mS/cm indicating a low total of dissolved salts, and concentrations of nitrogen, phosphorus, potassium, calcium, magnesium, and sulfur were all below 10 ppm. Total suspended solids (TSS, which is a measure of particles in the water) for the well water was also very low (less than 0.1 mg/L). The irrigation water had a slight orange-brown color, caused by the one notable contaminant: 1 ppm of dissolved iron.
In the greenhouse, the TSS increased to 6.3 mg/L, representing the particles that clogged the irrigation lines. With the addition of fertilizer nutrients, the pH was 5.6, EC was 1.9 mS/cm, and iron concentration was 1.9 ppm.
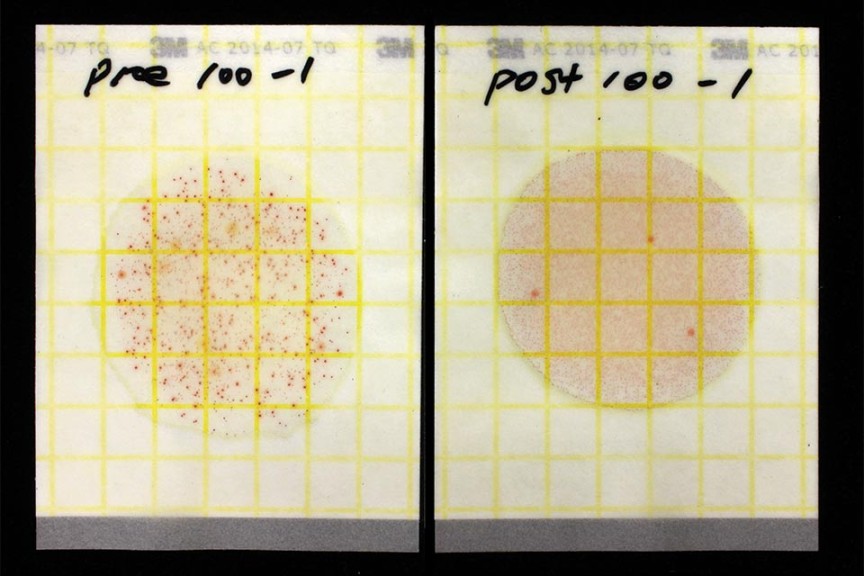
The University of Florida lab plated out aerobic bacteria in the water samples using 3M Petrifilms (Figure 3, see slideshow). Most local water quality laboratories can run a similar test for heterotrophic or aerobic bacteria counts. Bacterial counts in colony-forming units per milliliter (CFU/mL) increased from 48,300 at the well-head to 2,117,000 at the drip emitter. Levels above 5,000 CFU/mL with the Petrifilm test (or above 10,000 CFU/mL with most agar-based tests of water quality) indicate a high risk of biofilm. Therefore, measured bacteria counts were high at the well, and extremely high in the greenhouse.
The Culprit: Iron Bacteria
The level of particles (TSS) was very low in the well water, so it was not an initial silt, clay, or sand particle issue. Suspended solids were forming within the irrigation lines, resulting in a high TSS in the greenhouse. The low EC and ion levels meant mineral precipitation would not occur.
Bacteria counts were high at the well-head. Usually, well water has low levels of bacteria compared to sources such as ponds or rivers. However, ions including iron, manganese, or sulfur can sometimes be energy sources for bacteria to grow. Iron bacteria derive energy to live and multiply by oxidizing dissolved ferrous iron (Fe2+) to solid ferric oxide (Fe3+, forming rust particles). Iron levels in the citrus greenhouse were high enough (above 0.3 ppm) to cause bacteria issues.
The Solution
What can be done about this problem? There are different options to remove iron by oxidizing it (adding oxygen to turn the dissolved iron into solid rust), and killing the bacteria. This is followed by filtering out the rust particles.
In this case, the grower chose to install a chlorination system using liquid sodium hypochlorite (bleach) because it is low cost and requires a simple dosing pump to provide 2 ppm (Figure 4, see slideshow). The grower also purchased a free and total chlorine testing kit to check dosage and ensure that a small residual amount (0.4 ppm total chlorine) was still present out of the drippers.
Workers replaced the screen filter with two 36-inch sand filters in parallel, filled with #20 to #30 crushed silica, to improve removal of small iron particles. They placed the filter further down the irrigation line to allow for several minutes of chlorination before filtration. Fertilizer injection occurred after the iron removal process, because chlorine is more effective at sanitizing clear water than sanitizing a nutrient solution. The filters were back-flushed out to the ground daily to clean out particles. Back-flushing was triggered automatically by time or when a pressure gauge indicated a significant drop in pressure after the filter, which reduced the maintenance labor and improved system performance.
The Result
Looking at the back-flush water in Figure 4 (see slideshow), you can clearly see the iron particles removed by chlorination and filtration. We tested bacteria counts on another set of samples at the well and out in the greenhouse, which decreased from 56,700 CFU/mL at the well-head to zero after the filter and zero out in the greenhouse. Clogging has not been a problem after several months of operation, but monitoring will be ongoing.
Because of the crop losses and the extra labor from the biofilm problem, it was easy to estimate the benefits of unclogging the drip emitters (Table 2, see slideshow). Choosing low-cost technologies (chlorine and sand filtration) meant a rapid payback on this investment.
Take-Home Lessons
• If you have clogged emitters, identify which of the three common factors are causing the clogging.
• If biofilm is the issue, identify how bacteria levels are changing within the irrigation lines by testing bacteria count at different points in the system.
• You must treat microbial contamination occurring at the water source. If biofilm is building up within the lines, also provide some residual treatment that carries through to the emitters.
• Consider costs and benefits — evaluate simple, proven, and low-cost technologies first.
Thanks to funding support and collaboration from industry partners the Floriculture Research Alliance (FloricultureAlliance.org). This case study is part of the Clean WateR3 research project funded through the USDA-NIFA Specialty Crops Research Initiative. For more information, see CleanWateR3.org.