How to Prevent Petunia Flower Meltdown From Botrytis

Petunia flower meltdown caused by Botrytis cinerea infection. Photos by Jim Faust.
Botrytis cinerea is a common fungal pathogen that can be deadly to your plants. In the May 2018 issue of Greenhouse Grower, we discussed the results of evaluating fungicide resistance in Botrytis cinerea, the causal agent of gray mold disease. We demonstrated how resistance to fungicides can be a serious problem for the greenhouse industry around the world. Several years ago, with the financial support of the American Floral Endowment, we examined an array of alternative compounds that were believed to have some potential to control gray mold. This article will discuss the results that point to an alternative strategy for managing gray mold on highly susceptible flowering crops, such as petunia.
Background
In the greenhouse, Botrytis spores are always present, being picked up by air movement and landing on crop surfaces. The spores germinate when environmental conditions are favorable, for example, when the relative humidity is 94% and higher or when the duration of leaf wetness is 12 hours or more at moderate temperatures (64°F to 77°F). This environment may occur inside the greenhouse when the outdoor weather is cloudy and rainy. Conditions during shipping may also create an environment favorable for infection when the plants are densely packed on carts, the carts are wrapped in plastic, and the carts are not immediately unpacked upon delivery to the retail store.
The conventional approach to protecting plants from Botrytis infection is to make weekly fungicide applications. This, however, can lead to fungicide resistance development. We have shown that some of the spores collected from petunias grown in a commercial greenhouse were resistant to six common fungicides of different chemical classes, including fludioxonil, pyraclostrobin, boscalid, thiophanate-methyl, fenhexamid, and iprodione. Thus, we began a search for alternatives to conventional fungicides.
Calcium to the Rescue
After examining several different compounds, including a fungicide, we found that calcium chloride spray applications were providing the best Botrytis control. In fact, the calcium treatments were consistently equal to or better than the fungicide treatments. Calcium chloride was applied to petunias at varying rates (0, 400, 800 and 1,200 ppm Ca) twice a week for two weeks. Then, newly opened flowers were removed from the plants and placed in humid chambers. The flowers were then inoculated with a solution containing Botrytis spores, and the rate of disease infection severity was recorded.

Effect of calcium chloride spray applications on petunia flowers following inoculation with Botrytis spores.
For the last three decades, growers have been spraying poinsettias with 400 ppm calcium to prevent bract edge burn. Bract-edge burn symptoms are caused by localized calcium deficiencies along the bract margins that make the tissue more susceptible to Botrytis infection. The difference in our current work is that we are applying higher rates of calcium to help strengthen flower tissue to become more resistant to infections.
Phytotoxicity was not observed during this experiment, but we have seen some phytotoxicity when applying more than 1,000 ppm of calcium using calcium chloride on mature petunia flowers. Our data show that closed flower buds are more resistant to phytotoxicity. Phytotoxicity is a major concern, and there is differing sensitivity amongst varieties, species, and calcium formulations. Growers should only use this information as a starting point for trialing this technique in their own facilities.
How Does It Work?
Calcium movement into reproductive organs (e.g., flowers and fruit) can be low because calcium is transported mainly in the xylem while flowers are primarily supplied with nutrients through the phloem. This causes a significantly lower amount of calcium movement into flower petals, making the flowers more susceptible to Botrytis infection than the leaves.
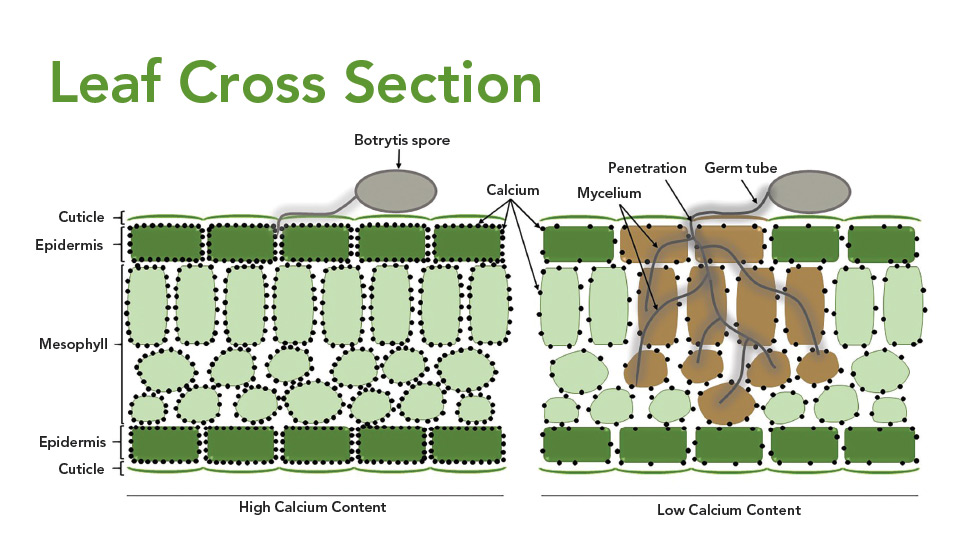
A Botrytis spore on the leaf surface produces a germ tube and then the mycelium attempts to feed on the contents of the plant cells by secreting an enzyme that aids in the digestion of the plant tissues. Left: Plant tissues with high calcium content resist Botrytis penetration into the cells. Right: Tissues with low calcium content allow the fungus to penetrate and kill the plant cells, resulting in necrotic (brown) tissues.
Calcium works by binding with the pectic material in cell walls, making it harder for the Botrytis to penetrate the cell and cause infection. More specifically, after the Botrytis spore germinates on the flower petal, it forms a germ tube that penetrates the cell wall of the plant tissue, and then forms mycelium that colonizes the plant tissue to feed on the contents of those cells (Figure 3, page 80). The fungus secretes an enzyme to help digest the cell wall of the host plant. Once infection occurs, the cell dies and the mycelium continues to the next cell. Higher calcium concentrations allow for more pectic bonds to be formed and improve the cell’s resistance to breakdown by the enzymes produced by Botrytis.
Comparing Foliar Sprays to Fertigation
Calcium can be supplied to plants in different ways, but it needs to be taken up by the flower petals to have an effect on Botrytis growth. For this reason, optimizing the delivery of calcium is critical. Preliminary results indicate that increasing calcium concentration in the fertigation solution will increase the calcium concentration within the leaves, but the calcium content in the flower petals does not increase as strongly. However, spray applications are quite effective for increasing the calcium concentration in the flowers. Unopened flower buds that are sprayed will continue to have higher calcium concentrations when they open.
Future Directions
The American Floral Endowment continues to support our research efforts exploring practical techniques to improve Botrytis management. Later in the year, we will be evaluating different calcium sources to determine which ones are best for increasing the tissue concentrations of calcium while minimizing the risk of phytotoxicity. The effect of adjuvants, surfactants, and penetrants on enhancing calcium concentration in the tissue will also be explored. This effort will also be expanded to other flowering species.








