Stop Thrips From Hiding in Your Greenhouse
Thrips are sneaky; they like to hide. Biocontrol programs against thrips start early in the crop cycle. Sprenches of nematodes, releases of predatory mites on the foliage and substrate, and when required, a few applications of a biopesticide like BotaniGard (BioWorks) should do the trick, right?
Not if you are carrying thrips into your greenhouse, without knowing it, every week.
Despite propagators’ best efforts, small numbers of pests may be hiding on cuttings, especially pests with widespread resistance to most conventional insecticides, such as western flower thrips (resistant to 30 active ingredients) and two-spotted spider mites (resistant to 95 active ingredients).
Thrips Hitchhike on Cuttings Far Too Often
To determine the frequency and the number of pests coming in this way, we sampled monthly chrysanthemum cuttings and also examined several batches of spring bedding plant cuttings.
Between July 2017 and March 2018, pot chrysanthemum cuttings were collected from local greenhouses before they were stuck. The majority (84%) of the samples contained thrips, and infestations were quite constant at around one to two thrips per batch of 20 cuttings. In addition, almost half of the cuttings (48%) had spider mites where the numbers were more variable and inconsistent, ranging from a few to 118 mites per 20 cuttings. Spider mite infestations occurred year-round, on all cultivars. Thrips were found mostly in the egg and larvae stages, in contrast to spider mites where all stages were observed.
Three batches of bedding plant cuttings (lantana, calibrachoa, ipomoea, verbena, impatiens, and osteospermum) were examined. In the first batch, no thrips were found. In the second one, we found thrips eggs and larvae on all plant species at a rate of one to two thrips per 20 cuttings. And in the third batch, only calibrachoa was infested with thrips (about 12 thrips per 20 cuttings), while the other plant species were clean.
From a few individuals, thrips populations in cuttings can increase to damaging levels within a few weeks, and early application of biocontrol agents may not be enough to keep up. It is also possible that residues on cuttings — from pesticides applied to stock plants — affect the efficacy of biocontrol agents during these early stages, resulting in a thrips outbreak.
Because it is extremely difficult and time-consuming to scout all incoming cuttings, it is probably easiest to assume that the cuttings are infested and take appropriate measures.
So, What Can You Do?
Previously, we found that cutting dips could reduce Bemisia whitefly infestations on poinsettia propagative materials to levels where they can easily be managed thereafter using biocontrol. Building on these results, we continued to see if applications of cutting dips containing reduced-risk materials, such as insecticidal soap (Kopa), oils (Landscape oil, SuffOil-X, Vegol), and biocontrol agents (BotaniGard 22WP, Nemasys) can reduce thrips infestations in a similar manner.
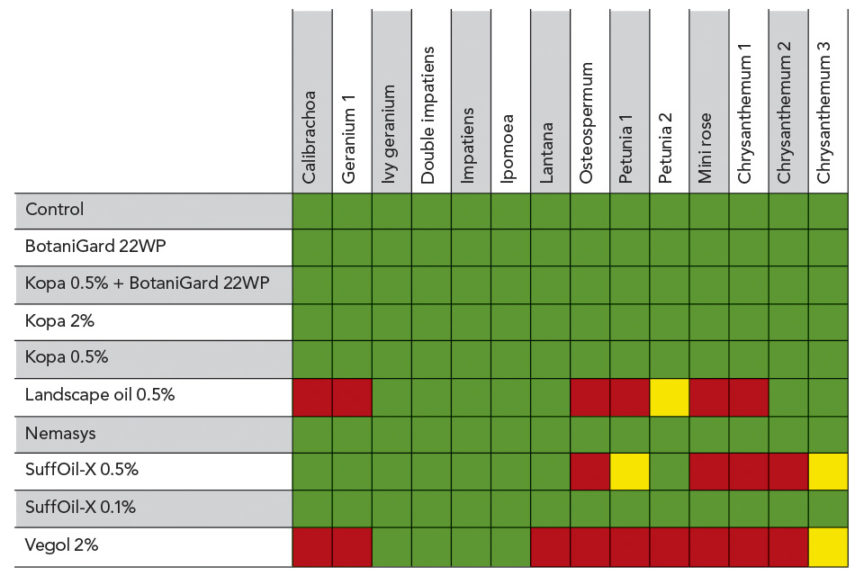
Most products and product combinations did not cause damage to a range of common crops, including several bedding plants, chrysanthemums, and mini-roses (see Table 1 and Figure 1 in slideshow). However, for certain plants the following dip combinations should be avoided: Landscape oil 0.5% (v/v), SuffOil-X 0.5%, and Vegol 2% were phytotoxic to certain plant species/varieties (see Table 1 in slideshow). The lower rate of SuffOil-X (0.1%) did not cause damage.
Cutting Dips Are Promising Tools to Help You Start Clean
Next, to determine the efficacy of these dips, we infested chrysanthemum cuttings with a mixed population of thrips eggs and larvae, dipped them, and incubated them for several days before assessing thrips survival (see Figure 2 and Figure 3 in slideshow).
Oil-based products were found to be the most effective dips and reduced thrips on cuttings by 75% to 95% compared to the control. Mineral oils (SuffOil-X and Landscape oil) were especially effective because they seem to kill both thrips eggs, which are laid in the leaf tissue, and thrips larvae. The challenge is now to find rates of oils that are not phytotoxic, but still effective against thrips.
Surprisingly, dips in Nemasys or BotaniGard 22WP (with or without Kopa) did not cause significant thrips mortality. It is likely that the principal thrips stages on the cuttings (i.e., eggs and larvae) are not very susceptible to nematodes, as was seen in previous research. In its traditional use as a foliar spray, BotaniGard 22WP is known to suppress thrips populations, so dip trials will be repeated to see if the experimental conditions affected the results. Another option may be to apply BotaniGard WP or Nemasys as a spray later in propagation. This application method is currently under investigation by our colleagues at Michigan State University.
In conclusion, thrips are definitely present on cuttings, and dips are a promising technique that can be easily implemented, allowing growers to start clean. In the coming year, we will validate results in greenhouse trials and work with registrants to include dipping as a legal application method on labels of the most successful products.