Pinpoint Toxicity In Your Pond Water
This month’s case study highlights a coastal Florida producer who was starting to use pond water for irrigation (Figure 1, see slideshow). The pond water had a lower electrical conductivity (EC) than the operation’s well water, and the well water supply was running low. However, the grower realized that the open pond had a high level of algae and could possibly harbor pathogens, so he installed a water-treatment system.
Water pumped from the pond passed through a sand filter, and sodium hypochlorite (liquid bleach) was injected to chlorinate the water. The water was batch-treated in a holding treatment tank before being pumped to the greenhouse, to ensure there was adequate contact time to control algae and pathogens (Figure 2, see slideshow).
Despite this treatment, plant health problems occurred when the pond water was applied in the greenhouse. Some plants showed pitting and bronzing symptoms on foliage, and stunted growth (Figure 3a and 3b, see slideshow).
Water Analysis Helps Narrow Down The Problem
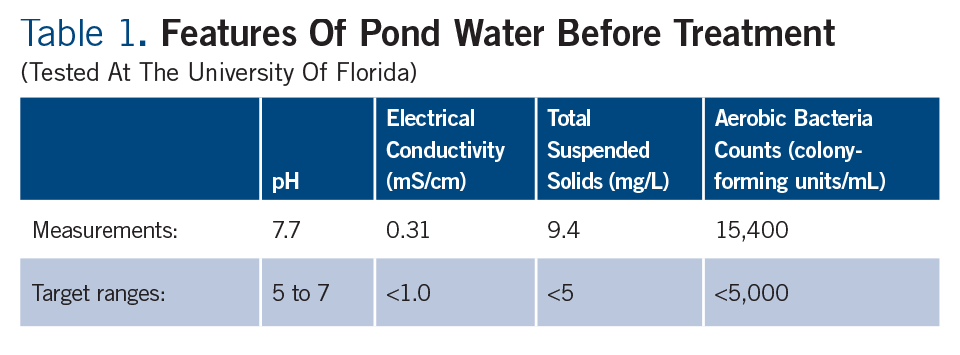
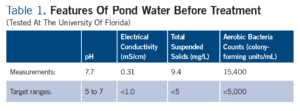
Given that the plant symptoms only occurred when the pond water was used, what could be the problem? A series of water tests were run on the untreated pond water (Table 1), but none of the results were far out of range:
• pH was slightly high, but plant symptoms were not consistent with iron deficiency at high pH.
• Concentration of dissolved salts (both electrical conductivity, and a detailed analysis of individual dissolved ions) was low. Applied fertilizer concentration in the greenhouse was also low, and pesticides were applied at normal label rates. Therefore, residue on foliage or high salts in the root substrate were not damaging plant health.
• Total suspended solids and bacteria counts were higher than desired, which is typical in pond water because of algae and microbial growth. However, the grower did not complain of clogged irrigation emitters after chlorination and filtration.
• No oomycetes (Pythium or Phytophthora) were found in the water sample, and plant symptoms were not consistent with root or crown rot.
That led us to focus on the chlorination treatment. The grower was applying 1.75 gallons of 10% chlorine in the 8,000-gallon tank. The test kit for free chlorine showed a reading of up to 3.5 ppm for the “residual” level coming out of the batch tank (see Table 2 for chlorine terms).
The Culprit: Overdosing Chlorine
Because the nutrients in the pond combined with the active ingredient of chlorine (hypochlorous acid), the total applied chlorine was much higher than the 3.5 ppm of free chlorine being read on the meter.
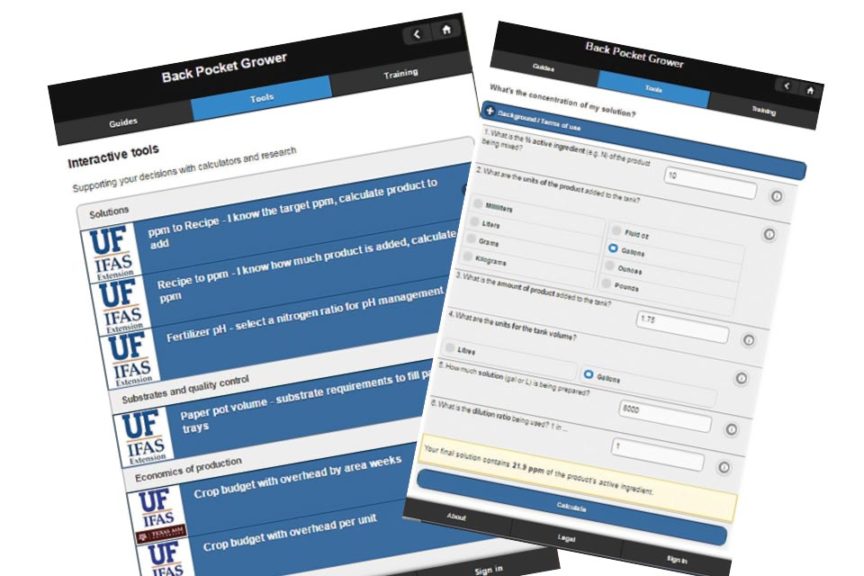
We calculated almost 22 ppm was being applied. This is much higher than the safe zone of 2 to 5 ppm of applied total chlorine, and 1 to 2 ppm of residual chlorine. To calculate the dose based on the gallons of bleach and tank size, go to our website BackPocketGrower.org, and under “Tools” use the “Recipe to ppm” tool (Figure 4, see slideshow).
We have run experiments in our greenhouse to test chlorine phytotoxicity. We have not seen damage when hypochlorous acid was applied overhead at 2 ppm with each irrigation to bedding plants. However, we have heard reports from commercial greenhouses where phytotoxicity occurs with low doses (2 ppm, especially in propagation). Chloramines, then hypochlorous acid, and then chlorine dioxide, in that order, have increasing reactivity and likelihood of phytotoxicity. We have also observed phytotoxicity from other sanitizing agents applied at high rates, including hydrogen dioxide-based products and copper ionization. Simply switching away from chlorine is therefore not always the answer.
Solution: Reduce Chlorine Dosage
The grower reduced the applied dose to no more than 3 ppm of total chlorine, and 1 to 2 ppm of residual chlorine. For safety, this was based on two ways to check dosage (a) measurements of total and free chlorine, and (b) calculated amount of chemical added per volume of irrigation water.
Take-Home Lessons
Most growers are not trained as water-treatment engineers, but many are familiar with technology not working. Like any piece of equipment, no water treatment system is maintenance-free. If you add a water treatment system to minimize risk from issues such as plant pathogens, realize that you are also adding risk (phytotoxicity or worker safety) from equipment malfunctioning.
• Avoid complicated equipment you do not understand and ensure that the supplier provides good service.
• Put a maintenance and monitoring standard operating procedure (SOP) into place. There is always drift away from the SOP, so regular checking by a supervisor is needed.
• Growers need to be involved in testing and interpreting the water treatment equipment. It is your crop in harm’s way, so do not just leave it to the physical plant staff.
• Do not rely just on the measured dose from a cheap testing kit. Check how much sanitizing agent is being used per unit volume of irrigation water. Put in an inline ORP (oxidation-reduction potential) sensor, which should read around 700 mV with 2 ppm free chlorine and pH 6.
• Never apply more than 5 ppm total chlorine, and never have more than 1 to 2 ppm of residual total chlorine. If the water demand is extremely high, then add filtration and sometimes flocculation upstream to reduce the demand, before chemical is added. Acidify the water to pH 6 so less chlorine is needed for the same sanitizing power.
Thanks to funding support and collaboration from industry partners the Floriculture Research Alliance (FloricultureAlliance.org). This case study is part of the Clean WateR3 research project funded through the USDA-NIFA Specialty Crops Research Initiative. For more information, see CleanWateR3.org.
In Case You Missed It
Unclog Drip Emitters In Your Greenhouse