Addressing pH Problems
If it was only as easy as turning a dial, pH problems would not exist. In the real world, the simplest way to avoid improper substrate pH is by keeping a close eye. Regularly monitoring substrate allows growers to identify when pH is just beginning to get out of range.
In most cases, small pH adjustments (0.2 to 0.6 pH units) are easily accomplished by simply changing the type of fertilizer. Large pH adjustments can be time consuming, costly and typically result from infrequent pH monitoring.
The green line in Figure 1 (see page 44) represents a situation in which a grower is monitoring marigold substrate pH on a weekly basis. At week 4, the substrate pH has fallen out of the ideal pH zone and into the small corrective zone. The grower may have been using a fertilizer too acidic for the situation, and simply switching to a more basic fertilizer causes the pH to increase back into the ideal pH zone by week 6.
The red line represents a situation in which pH is not monitored and low pH problems are most likely noticed as a manifestation of micronutrient toxicity in the plants. Micronutrient toxicity is a common problem caused by low pH. In the latter situation, the pH is too low by week 6 to be restored with a switch to a more basic fertilizer.
To solve this pH problem, growers will have to spend a lot of time mixing a solution of flowable lime or potassium bicarbonate, applying the solution to the crop and then washing off the foliage. This situation could easily be avoided with frequent pH monitoring and a simple fertilizer switch.
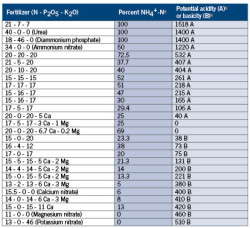
The form of nitrogen in your fertilizer is what causes substrate pH to decrease or increase. Nitrogen is the most important pH-controlling ion because it is the only element required by plants that can be supplied as both a positive cation (ammonium: NH4+) or a negative anion (nitrate: NO3–) and accounts for more than half of the nutrient ions taken up by the plant. Fertilizers high in ammonium have an acidifying effect and cause substrate pH to decrease, and the opposite is true for fertilizers high in nitrate (Table 1).
When ammonium (or other positive cations) is taken up by the plant, a positive charge enters the root. Plants must remain electrochemically neutral, and thus the roots secrete positively charged H+, which reduces the pH (Figure 2). When nitrate (or other negative anions) is absorbed, the roots balance the negative charge by absorbing H+. As more nitrate is absorbed, more H+ is removed from the soil solution, and the substrate pH increases (Figure 3, see page 46).
Urea is a third form of nitrogen that is a common component of fertilizer. Once in the soil or the plant, urea is split into carbon dioxide and ammonium by the urease enzyme produced by microorganisms or the plant. For this reason, urea is considered equivalent to ammonium in respect to effect on substrate pH.
Ammonium Toxicty
When using fertilizers high in ammonium or urea, there is the threat of ammonium toxicity. Plants have the ability to store large amounts of nitrate but do not have this ability for ammonium, therefore excess uptake is toxic. If one was supplying less than or equal to the exact amount of nitrogen required by the plant, the ammonium plus urea could be 100 percent of the nitrogen.
However, in most greenhouse situations, we are supplying nitrogen in excess of the plants’ needs. Another consideration when using fertilizers high in ammonium plus urea are nitrifying bacteria. These soil microorganisms convert ammonium to nitrate and are much less active at low temperatures, low-oxygen concentrations (saturated soil) and low pH. When temperatures and light level are low during the winter months, there is a greater risk of ammonium toxicity.
Typically problems will not occur when ammonium plus urea are less than 40 percent, and this percentage can be higher when conditions favor nitrifying bacteria.
Ammonium-To-Nitrate Ratio Research
The following research was conducted at the Institute for Horticulture of the Weihenstephan-Triesdorf University of Applied Sciences. The institute is north of Munich, which is an area with limestone bedrock and water alkalinity that can reach 300 ppm calcium carbonate equivalent (CCE). Water with this level of alkalinity or bicarbonate would typically need to be blended with rain water, treated with acid or filtered by reverse osmosis to be suitable for crop production. Water alkalinity is essentially dissolved limestone and if not treated, will lead to problems with high pH such as micronutrient deficiency.
If the concentration of alkalinity in water is zero, the opposite will occur and substrate pH will decrease over time. Plant roots and associated microorganisms are continually releasing carbon dioxide (CO2) as a byproduct of respiration. Carbon dioxide combines with water in the soil solution to form carbonic acid, which lowers substrate pH.
The purpose of the research was to determine if certain ammonium-to-nitrate ratios in fertilizer could offset the pH effect of highly alkaline water or water with zero alkalinity with calibrachoa ‘Superbells Royal Blue.’ Treatments were deionized water and tap water in combination with five fertilizer ammonium to nitrate ratios (90:10, 70:30, 50:50, 30:70 and 10:90).
When plants were grown with deionized water, substrate pH decreased over time regardless of fertilizer treatment. Generally, end-of-crop substrate pH increased as the amount of ammonium in the fertilizer decreased.
When plants were grown with high-alkalinity tap water, the opposite occurred and substrate pH increased over time, regardless of fertilizer treatment. Fertilizer treatment did not have as strong an effect on end-of-crop substrate pH as with deionized water. Results indicate the high alkalinity is the primary factor controlling the change in substrate pH. One bicarbonate ion (HCO3–) added to the soil solution with irrigation water will neutralize one H+ ion (Reaction 1). Reaction 2 shows the carbonic acid is then converted to H2O and CO2. This is the opposite of what was discussed above where CO2 and H2O react to form carbonic acid. With high amounts of bicarbonate in the system, the reactions will be pushed to the right and acid will be neutralized. Again, this is similar process whereby limestone neutralizes H+.
Reaction 1: H+ + HCO3– (bicarbonate) –> H2CO3 (carbonic acid) [H+ neutralized]
Reaction 2: H2CO3 –> H2O (water) + CO2 (carbon dioxide)
This research indicates when water alkalinity is extremely low or extremely high, adjusting the ammonium-to-nitrate ratio is not an effective method to completely offset the pH effect. The key to successful pH management is continual monitoring and having several types of fertilizer on hand to make small corrections when needed.
2
2
5
Addressing pH Problems










